I. Introduction: What are stem cells, and why are they important?
Stem cells have the remarkable potential to renew themselves. They can develop into many different cell types in the body during early life and growth. Researchers study many different types of stem cells. There are several main categories: the “pluripotent” stem cells (embryonic stem cells and induced pluripotent stem cells) and nonembryonic or somatic stem cells (commonly called “adult” stem cells). Pluripotent stem cells have the ability to differentiate into all of the cells of the adult body. Adult stem cells are found in a tissue or organ and can differentiate to yield the specialized cell types of that tissue or organ.
Pluripotent stem cells
Early mammalian embryos at the blastocyst stage contain two types of cells – cells of the inner cell mass, and cells of the trophectoderm. The trophectodermal cells contribute to the placenta. The inner cell mass will ultimately develop into the specialized cell types, tissues, and organs of the entire body of the organism. Previous work with mouse embryos led to the development of a method in 1998 to derive stem cells from the inner cell mass of preimplantation human embryos and to grow human embryonic stem cells (hESCs) in the laboratory. In 2006, researchers identified conditions that would allow some mature human adult cells to be reprogrammed into an embryonic stem cell-like state. Those reprogramed stem cells are called induced pluripotent stem cells (iPSCs).
Adult stem cells
Throughout the life of the organism, populations of adult stem cells serve as an internal repair system that generates replacements for cells that are lost through normal wear and tear, injury, or disease. Adult stem cells have been identified in many organs and tissues and are generally associated with specific anatomical locations. These stem cells may remain quiescent (non-dividing) for long periods of time until they are activated by a normal need for more cells to maintain and repair tissues.
II. What are the unique properties of all stem cells?
Stem cells have unique abilities to self-renew and to recreate functional tissues.
Stem cells have the ability to self-renew.
Unlike muscle cells, blood cells, or nerve cells—which do not normally replicate— stem cells may replicate many times. When a stem cell divides, the resulting two daughter cells may be: 1) both stem cells, 2) a stem cell and a more differentiated cell, or 3) both more differentiated cells. What controls the balance between these types of divisions to maintain stem cells at an appropriate level within a given tissue is not yet well known.
Discovering the mechanism behind self-renewal may make it possible to understand how cell fate (stem vs. non-stem) is regulated during normal embryonic development and post-natally, or misregulated as during aging, or even in the development of cancer. Such information may also enable scientists to grow stem cells more efficiently in the laboratory. The specific factors and conditions that allow pluripotent stem cells to remain undifferentiated are of great interest to scientists. It has taken many years of trial and error to learn to derive and maintain pluripotent stem cells in the laboratory without the cells spontaneously differentiating into specific cell types.
Stem cells have the ability to recreate functional tissues.
Pluripotent stem cells are undifferentiated; they do not have any tissue-specific characteristics (such as morphology or gene expression pattern) that allow them to perform specialized functions. Yet they can give rise to all of the differentiated cells in the body, such as heart muscle cells, blood cells, and nerve cells. On the other hand, adult stem cells differentiate to yield the specialized cell types of the tissue or organ in which they reside, and may have defining morphological features and patterns of gene expression reflective of that tissue.
Different types of stems cells have varying degrees of potency; that is, the number of different cell types that they can form. While differentiating, the cell usually goes through several stages, becoming more specialized at each step. Scientists are beginning to understand the signals that trigger each step of the differentiation process. Signals for cell differentiation include factors secreted by other cells, physical contact with neighboring cells, and certain molecules in the microenvironment.
III. How do you culture stem cells in the laboratory?
How are stem cells grown in the laboratory?
Growing cells in the laboratory is known as “cell culture.” Stem cells can proliferate in laboratory environments in a culture dish that contains a nutrient broth known as culture medium (which is optimized for growing different types of stem cells). Most stem cells attach, divide, and spread over the surface of the dish.
The culture dish becomes crowded as the cells divide, so they need to be re-plated in the process of subculturing, which is repeated periodically many times over many months. Each cycle of subculturing is referred to as a “passage.” The original cells can yield millions of stem cells. At any stage in the process, batches of cells can be frozen and shipped to other laboratories for further culture and experimentation.
How do you “reprogram” regular cells to make iPSCs?
Differentiated cells, such as skin cells, can be reprogrammed back into a pluripotent state. Reprogramming is achieved over several weeks by forced expression of genes that are known to be master regulators of pluripotency. At the end of this process, these master regulators will remodel the expression of an entire network of genes. Features of differentiated cells will be replaced by those associated with the pluripotent state, essentially reversing the developmental process.
How are stem cells stimulated to differentiate?
As long as the pluripotent stem cells are grown in culture under appropriate conditions, they can remain undifferentiated. To generate cultures of specific types of differentiated cells, scientists may change the chemical composition of the culture medium, alter the surface of the culture dish, or modify the cells by forcing the expression of specific genes. Through years of experimentation, scientists have established some basic protocols, or “recipes,” for the differentiation of pluripotent stem cells into some specific cell types.
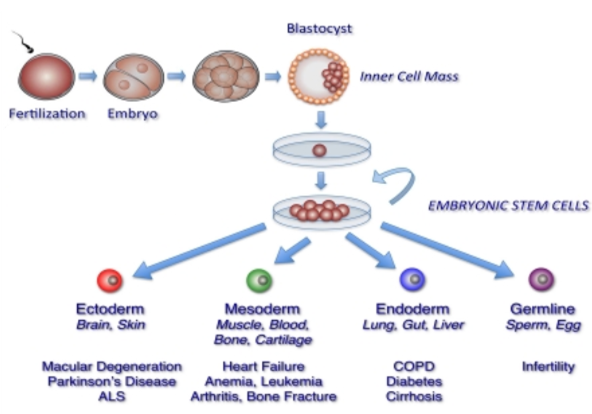
Adapted with author permission from “The promise of human embryonic stem cells in aging-associated diseases” by O. Yabut and H.S. Bernstein, 2011.
What laboratory tests are used to identify stem cells?
At various points during the process of generating stem cell lines, scientists test the cells to see whether they exhibit the fundamental properties that make them stem cells. These tests may include:
- Verifying expression of multiple genes that have been shown to be important for the function of stem cells.
- Checking the rate of proliferation.
- Checking the integrity of the genome by examining the chromosomes of selected cells.
- Demonstrating the differentiation potential of the cells by removing signals that maintain the cells in their undifferentiated state, which will cause pluripotent stem cells to spontaneously differentiate, or by adding signals that induce adult stem cells to differentiate into appropriate cell phenotypes.
IV. How are stem cells used in biomedical research and therapies?
Given their unique regenerative abilities, there are many ways in which human stem cells are being used in biomedical research and therapeutics development.
Understanding the biology of disease and testing drugs
Scientists can use stem cells to learn about human biology and for the development of therapeutics. A better understanding of the genetic and molecular signals that regulate cell division, specialization, and differentiation in stem cells can yield information about how diseases arise and suggest new strategies for therapy. Scientists can use iPSCs made from a patient and differentiate those iPSCs to create “organoids” (small models of organs) or tissue chips for studying diseased cells and testing drugs, with personalized results.
Cell-based therapies
An important potential application is the generation of cells and tissues for cell-based therapies, also called tissue engineering. The current need for transplantable tissues and organs far outweighs the available supply. Stem cells offer the possibility of a renewable source. There is typically a very small number of adult stem cells in each tissue, and once removed from the body, their capacity to divide is limited, making generation of large quantities of adult stem cells for therapies difficult. In contrast, pluripotent stem cells are less limited by starting material and renewal potential.
To realize the promise of stem cell therapies in diseases, scientists must be able to manipulate stem cells so that they possess the necessary characteristics for successful differentiation, transplantation, and engraftment. Scientists must also develop procedures for the administration of stem cell populations, along with the induction of vascularization (supplying blood vessels), for the regeneration and repair of three-dimensional solid tissues.
To be useful for transplant purposes, stem cells must be reproducibly made to:
- Proliferate extensively and generate sufficient quantities of cells for replacing lost or damaged tissues.
- Differentiate into the desired cell type(s).
- Survive in the recipient after transplant.
- Integrate into the surrounding tissue after transplant.
- Avoid rejection by the recipient's immune system.
- Function appropriately for the duration of the recipient's life.
While stem cells offer exciting promise for future therapies, significant technical hurdles remain that will likely only be overcome through years of intensive research.
Note: Currently, the only stem cell-based products that are approved for use by the U.S. Food and Drug Administration (FDA) for use in the United States consist of blood-forming stem cells (hematopoietic progenitor cells) derived from cord blood. These products are approved for limited use in patients with disorders that affect the body system that is involved in the production of blood (called the “hematopoietic” system). These FDA-approved stem cell products are listed on the FDA website. Bone marrow also is used for these treatments but is generally not regulated by the FDA for this use. The FDA recommends that people considering stem cell treatments make sure that the treatment is either FDA-approved or being studied under an Investigational New Drug Application (IND), which is a clinical investigation plan submitted and allowed to proceed by the FDA.
V. How does NIH support stem cell research?
NIH conducts and funds basic, translational, and clinical research with a range of different types of stem cells. NIH-supported research with human pluripotent stem cells is conducted under the terms of the NIH Guidelines for Human Stem Cell Research. NIH awards are listed in various categories of stem cell research through the NIH Estimates of Funding for Various Research, Condition, and Disease Categories (RCDC). NIH also supports a major adult stem cell and iPSC research initiative through the Regenerative Medicine Innovation Project.